These TS 10th Class Physical Science Chapter Wise Important Questions Chapter 7 Classification of Elements- The Periodic Table will help the students to improve their time and approach.
TS 10th Class Physical Science Important Questions Chapter 7 Classification of Elements- The Periodic Table
1 Mark Questions
Question 1.
Define element’ according to Boyle.
Answer:
Robert Boyle defined an element as any substance that cannot be decomposed into a further simple substance by a physical or chemical change.
Question 2.
State law of triads.
Answer:
Dobereiner stated that when elements with similar properties are taken three at a time and arranged in the ascending order of their atomic weights the atomic weight of the middle element Is the average of the atomic weights of the first and third elements.
Question 3.
What are the limitations of Doberelner’s law of trade?
Answer:
- All the known elements then could not be arranged in the form of triads.
- The law failed for very low mass or for very high mass elements.
- As the techniques improved for measuring atomic masses accurately, the law was unable to remain strictly valid.
Question 4.
State the law of octaves.
Answer:
The law of octaves states that when elements are arranged In the ascending order of their atomic weights, they fall Into a pattern in which their properties repeat at regular Intervals. Every 8th element starting from a given element resembles In Its properties to that of the starting element. This is similar to octve notes In musical scale.
![]()
Question 5.
What are the limitations for Newlands’ law of octaves?
Answer:
- There are instances of two elements fitted into the same slot. Eg: Co and Ni.
- Certain elements totally are dissimilar n their properties, were fitted into the same group.
- The law was not valid for elements that had atomic masses higher than that of Calcium.
- It was restricted to 56 elements and did not leave any room for new elements.
Question 6.
What is Doberelner triad ? Give two examples to It.
Answer:
A group of three elements In which atomic weight of middle element is average of first and third element is called Dobereiner triad.
Eg:
1) Li,Na,K
2) Cl,Br,I
Question 7.
What is Mendeleeffs periodic law?
Answer:
Mendeleeffs periodic law: The physical and chemical properties of the elements are the periodic functions of their atomic weights.
Question 8.
What is valency?
Answer:
The combining power of elements with respect to hydrogen, oxygen or indirectly any other element through hydrogen and oxygen is called valency.
Question 9.
What is modern periodic law?
Answer:
Modern periodic law: The properties of elements are periodic functions of their atomic numbers.
Question 10.
What is the name given to 1(A) group elements?
Answer:
Alkali metal family, because aliqulli plant ashes. Na; K etc., were obtained from plant ash.
Question 11.
What are halogens?
Answer:
Fluorine, Chlorine, Bromine, Iodine, and Astatine of VII A group elements are called halogens, which are obtained from sea salt.
![]()
Question 12.
What are noble gases?
Answer:
The elements of group VIII A(18) are chemically least reactive. So they are called as noble gases. Their group electronic configuration ns2np6 (except) for helium it is Is2.
Question 13.
What are Lanthanides?
Answer:
Elements acquiring same properties are called lanthanides i.e., 4f elements. They are from 58Ce(Cenum) to 71Lu(Lutetium).
Question 14.
What are Actinides?
Answer:
Elements acquiring different properties are called actinides i.e., 5f elements. They are from 90Th(Thorium) to 103 Lr(Lawrencium).
Question 15.
What are metals and non-metals?
Answer:
The elements with three or less electrons in the outer shell are considered to be metals and the ones with five or more electrons in the outer shell are considered to be non-metals.
Question 16.
What is electronegativity?
Answer:
The relative tendency of its atom to attract electrons towards Itself when it is bounded to the atom or another element is called electronegativity.
Question 17.
What is electropositive character?
Answer:
The tendency of metals to remain positive Ions in compounds is called electropositive character.
Question 18.
What are metalloids?
Answer:
Metalloids or semi-metals are elements which have properties that are intermediate between the properties of metals and non-metals. They possess properties like metals but brittle like non-metals. They are generally semiconductors. Eg: B, Si, Ge, etc.
Question 19.
Why the elements in a group have similar chemical properties?
Answer:
Physical and chemical properties of elements are related to their electronic configurations, particularly the outer shell configurations. The atoms of the elements in a group possess similar electronic configurations. Therefore we expect all the elements In a group should have similar chemical properties.
Question 20.
Why the elements In a period possess different chemical properties?
Answer:
Across the table i.e. from left to right in any period elements get an increase in atomic number by one unit between any two successive elements. Therefore, the electron configuration of valence shell of any two elements in a given period is not same. Due to this reason, elements along a period possess different
chemical properties.
Question 21.
Define atomic radius.
Answer:
Atomic radius is the distance between the centre of the atomic nucleus and the electron cloud of the outermost energy level.
Question 22.
What is crystal radius (or) metallic radius?
Answer:
Half o, the distance between the centres of the nudei of two adjacent metal atoms In the metallic crystal is called crystal radius or metallic radius.
![]()
Question 23.
What is covalent radius?
Answer:
Covalent radius is half of the distance between the nuclei of two atoms, held together by a covalent bond In a homo-atomic molecule.
Question 24.
How does the atomic radius vary in a group and a period?
Answer:
In a group as we go down, the atomic radius Increases, and In a period, as we move from left to right, the atomic radius decreases.
Question 25.
Define IonIzation energy.
Answer:
The energy required to remove an electron from the outermost orbit or shell of a neutral gaseous atom is called Ionization energy.
Question 26.
How does the Ionization energy vary In group and period?
Answer:
In a group as we move down, Ionization energy decreases and in a period, as we move from left to right, the Ionization energy increases, but does not follow gradient order.
Question 27.
Define electron affinity.
Answer:
The electron affinity of an element is defined as the energy liberated when an electron Is added to its neutral gaseous atom. Electron affinity of an element is also known as electron gain enthalpy of that element.
Question 28.
How does the electron gain enthalpy values vary in a group and a period?
Answer:
Electron gain enthalpy values decrease as we go down in a group and increase along a period from left to right.
Question 29.
What is the Milliken’s proposal about electronegativity?
Answer:
Milliken proposed that the electronegativity of an element is the average value of its ionization energy and electron affinity.
Electronegativlty = \(\frac{\text { Ionization energy }+\text { Electron affinity }}{2} \)
Question 30.
How do the electronegativity vary In a group and in a period?
Answer:
Electronegativity values of elements decrease as we go down in a group and increase along a period from left to right.
Question 31.
How does metallic and non-metallic characters vary In a group and period?
Answer:
- Metallic character increases while non-metallic character decreases in a group as we move from top to bottom.
- Metallic character decreases while non-metallic character increases in a period as we move from left to right.
Question 32.
Give example(s) where the electronic configuration of an element does not JustIfy Its Inclusion In a block of element.
Answer:
- He (1s2) is Included in p-block elements.
- H (1s1) is not given any specific place. It can be also studied along with p- block halogens.
Question 33.
How many elements are present In the 5th period of the long-form periodic table? Give a possible reason.
Answer:
The 5th period starts with the filling of 5s level. It ends when the 5p level Is complete. The sub-energy levels filled in the sequence are 5s, 4d, 5p. Then the total number of electrons filled into these levels is 18. Hence the total number of elements in the period Is 18. The period starts with Rubidium (Rb) and ends with Xenon (Xe).
![]()
Question 34.
What do you mean by screening effect?
Answer:
The influence of the inner core of electrons on the attraction of the nucleus towards outer electrons is referred as “shielding effect’ or “screening effect”.
Question 35.
Arrange the elements B, N, Be and O in the increasing order of their ionizatIon potentials.
Answer:
The increasing order of ionization energy of B, N, Be and O is 0< N
Question 36.
What is meant by first Ionization energy?
Answer:
The energy required to remove first electron from the outermost orbit or shell of a neutral gaseous atom Is called first Ionization energy (1E1)
Question 37.
What is meant by second Ionization energy?
Answer:
The energy required to remove an electron from a uni-positive ion is called the second Ionization energy (1E2).
Question 38.
What do you mean by element family?
Answer:
Group of elements Is also called element family or chemical family. Eg: Group 1 (IA) is from Li to Fr with outer shell configuration ns ‘ad is called Alkali metal family.
Question 39.
Give the outer orbits’ general electronic configuration of the following types of elements, a) Noble gases b) Representative elements c) Transition elements d) Inner transition elements.
Answer:
The general electronic configuration of
(a) Noble gas is ns2 np6 except He. For He: 1s2
(b) Representative elements
s – block: ns1 and ns2
p – block : ns2 np1 to ns2 np5.
(c) Transition elements : (n – 1) d(0 -10) ns1or2 (where n ≥ 4)
(d) Inner transition elements : (n – 2) f1 to 14 (n – 1) d(1 to 6) ns2.
Question 40.
Chlorine, bromine, iodine are Doberelner’s triads. How do you justify?
Answer:
Chlorine, bromine and Iodine have similar properties and atomic weight of bromine Is average of chlorine and iodine.
Question 41.
Why are lanthanoids and actinoids placed separately at the bottom of the periodic table?
Answer:
Lanthanoids and actinoids belong to f – block elements with different properties so they placed at the bottom of periodic table.
Question 42.
Second Ionization energy of an element Is higher than Its first Ionization energy Why?
Answer:
It is difficult to remove an electron from unipositive ion when compared with neutral atom. So second ionization energy is always greater than first ionization energy.
Question 43.
Hydrogen can be placed in group 1. and group 7 periodic tables. Why?
Answer:
Hydrogen has both +1 as well as -1 oxidation states, So still, there is some ambiguity in position of hydrogen.
Question 44.
A, B are two elements. Its compound material Is A,B, then what will be the valency of A and B?
Answer:
The valency of A Island B is 2.
2 Marks Questions
Question 1.
Referring the part of the periodic table given below answer the questions that follow.
| Li | Be | B | C | N | O | F |
| Na | Mg | Al | Si | P | S | Cl |
a) What happens to the atomic size If moved from left to right? Support your answer.
b) What changes do you observe In the metallic properties of the elements when moved from left to right?
Answer:
a) When we move from left to right in a periodic table atomic radii of elements decrease, as a result the size of the atom decreases.
b) When we move From left to right in a periodic table electronegativity values of elements decrease, as a result the metallic properties of the elements increase.
![]()
Question 2.
The second-period element ‘F’ has electron gain enthalpy than the third-period elements of same group ‘ci’. Why?
Answer:
In a group of elements, the electron gain enthalpy decreases from top to bottom. But in general the second element in a group i.e., period element has greater electron gain enthalpy than the first element i.e. 2nd-period element.
Eg: E.An of F
This Is because Fluorine atom is smaller in size than chlorine atom. F2 is also having strong Inter electronic repulsions. In the addition of an electron to fluorine atom, the electronic repulsions overcome at the expense of a part of the energy liberated. Hence the overall energy lIberated Is less than that of chlorine atom.
Question 3.
x, y, and z are the elements of a Dobereiner’s triad. If the atomic mass of ‘z’ Is 7 and that of ‘Z’ In 39. What should be the atomic mass of the ‘Y’?
Answer:
The atomic mass of x = 7
The atomic mass of z = 39
x, y, z form Dobereiner triad
Atomic mass of y = average of x and z
= \(\frac{7+39}{2}=\frac{46}{2}\) = 23
Question 4.
Differentiate the metals and non-metals.
Answer:
| Metals | Non-Metals |
| 1. Metals have lustrous surfaces. | 1. Non-metals do not have lustrous surface |
| 2. They show malleability | 2. They do not show malleability |
| 3. They show ductility. | 3. They do not show ductility. |
| 4. They produce sonorous sounds. | 4. They do not produce sonorous sound |
| 5. Generally they are hard. | 5. Generally they are soft. |
| 6. They are good conductors of electricity. | 6. Generally they are bad conductors of electricity. |
| 7. Generally they liberate hydrogen gas when they are treated with acids. | 7. They do not liberate hydrogen gas. |
Question 5.
How are the elements divided into s, p, d, and f – blocks in the Modern periodic table?
Answer:
Depending upon to which sub-shells the differentiating electron i.e., the last coming electron enters in the atom of the given element, the elements are classified as s, p, d, and f-block elements. Eg: 11Na.
The electronic configuration of Na is 1s22s22p63s1.
The differentiating electron gets into 3s level, Therefore 11Na belongs to s block. Similarly 13Al(1S22s22p63s23p1) belongs to p-orbital
21Sc (1s22s22p63s23p64s23d1) belongs to d-block and
58Ce (1s22s22p63s23p64s23d104p64d105s25p66s24f1) belongs to f-block.
Question 6.
Explain the limitations of Mendeleefrs’ periodic table.
Answer:
1. Anomalous pair of elements: Certain elements of highest atomic weights precede those with lower atomic weights. Eg: TellurIum (At.wt. 127.6) precedes Iodine (at. wt. 126.9).
2. Dissimilar elements placed together: Elements with dissimilar properties were placed in same group as sub-group A and sub-group B.
Eg:
1. Alkali metals like U, Na, K, etc.. of IA group have little resemblance with coinage metals like Cu, Ag, Au of lB group.
2. Cl is of VIIA group and ‘Mn’is of VII B. But chlorine Is a non-metal where as Mn is a metal.
![]()
Question 7.
Observe the Information provided in the table and answer the questions given below It.
| Element | Na | C | Ca | P | Ti | Ni |
| Atomic Number | 11 | 6 | 20 | 15 | 22 | 28 |
(i) What are the s-block elements in the table?
(ii) What are the ‘p’ block and ‘d’ block elements In the table?
Answer:
(i) Na,Ca
(ii) C. P are p-block elements
Ti, Ni are d-block elements.
4 Marks Questions
Question 1.
Some elements belonging to second period of periodic table, and their atomic radii are given below. Observe them and write answers.
| 2nd period elements | B | Be | O | N | Li | C |
| Atomic radii | 88 | 111 | 66 | 74 | 152 | 77 |
a) Write the elements in the ascending order of their atomic radii.
b) Which of the 2nd-period elements closer to the configuration of Inert gas?
C) Which is the outermost orbit of all these elements?
d) Which element’s atomic size bigger, Beryllium or Carbon? Why?
Answer:
a) The ascending order of atomic sizes is O. N, C. B, Be and Li.
b) Lithium has closest inert gas configuration i.e.. 1S22st. Its nearest inert gas is Helium.
c) The outermost orbit for all these elements is second orbit.
d) Beryllium has more atomic size than Carbon. Because when we move across a period the atomic number increases. So nuclear attraction of outermost orbit increases. So carbon has lesser size than Beryllium.
Question 2.
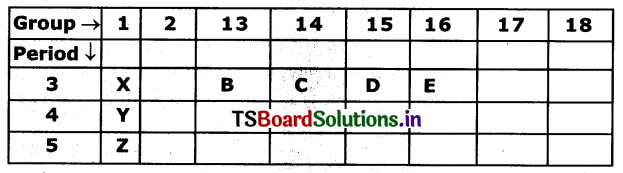
Refer the above part of periodic table and answer the following questions.
a) Element with the least atomic size.
b) Write th, electronic configuration of the elements B and E.
c) Identify the elements that have similar physical and chemical properties as the element Y.
d) Arranged elements increasing order of their electro-negativity values.
Answer:
a) The element with least atomic size is E. Because when we move from left to right in a period the atomic size decreases.
b) Electronic configuration of B is 1s22s22p63s23p1. Because the element belongs to 13th group its general configuration is ns2nP1 and the element belongs to third period and its atomic number is 13. Similarly electronic configuration of E ¡s 1s22s22p63s23p4. Because the element belongs to 16th group. Its general configuration is ns2np4 and third period. So its atomic number is 16.
c) The elements whid, have similar physical and chemical properties with Y are X and Z. Because they lie in a single group that Is l group. In a group, elements are having similar physical and chemical properties.
d) Z has highest atomic size because In a group atomic size Increases.
Question 3.
The electronic configuration of atom A is 2, 8, 6
a) What is the atomic number of element A?
b) State whether the atomic size of element A Is bigger or smaller than the atom having atomic number 14. Why?
c) Which of the elements exhibit similarity in chemical properties as element A 0(8), C(6), N(7), AV(1.8). Why?
d) How the element is formed Inert gas configuration?
Answer:
The electronic configuration of atom – A is 2, 8, 6.
a) Atomic number of element ‘A’ Is 16. i.e., Sulphur.
b) The atom which having atomic number – 14 is Silicon (SI). Atomic size of element decreases across periods from left to right. So the atomic size of element ‘A’ is smaller than the atom having atomic number 14.
c) Element oxygen O – exhibits similarity in chemical properties as element A, because they belong to the same group.
d) Given element – A becomes inert gas i.e., Argon configuration by gaining electrons.
![]()
Question 4.
Mendeleeff classified the then-known 63 elements in the form of a periodic table. Mention any two things that benefitted study of chemistry, to support above statement.
Answer:
- Mendeleeff accepted minor inversions in the order of increasing atomic weights as these inversions resulted in elements being placed in the correct group.
- It was the extraordinary thinking of Mendeleeff that made the chemists to accept the periodic table and recognize Mendeleeff more than anyone else as the originator of the periodic law.
- At the time when Mendeleeff introduced his periodIc table, even electrons were not discovered.
- Even then the periodic table provides a scientific base for the study of chemistry of elements.
- In his honour the 101st element was named “Mendelevlum’
Question 5.
Explain the salient features and achievements of the Mendeleeff’s periodic table.
Answer:
Mendeleeff arranged the elements in the increasing order of their atomic weights.
Salient Features:
1. Groups and sub-groups: There are 8 vertical columns called groups. represented by Roman numerals Ito VIII. Each group s divided into two subgroups A and B. The elements present in a group and its subgroup have similar properties.
2. Periods: The horizontal rows In Mendeleeff’s table are caned ‘periods’. There are 7 periods, denoted by Arab numerals I to 7. A period comprises the entire range of elements after which properties repeat themselves.
3. Predicting the properties of missing elements: Based on the arrangement of the elements in the table, he predicted that some elements were missing and left blank spaces at the appropriate places in the table. He named those elements tentatively by adding the prefix ‘eka’ to the name of the element immediately above the each empty space. Eg: Eka-boron, Eka – aluminum, Eka – silicon were close to the observed properties of Scandium, Galium and Germanium respectively which were discovered later.
4. Correction of atomic weight: The correct placement of elements In Mendeleeff’s periodic table helped in correcting the atomic masses of some elements like Beryllium, Indium, Gold.
5. Anomalous series: Some anomalous series of elements like ‘Te’ and I were observed in the table. The anomalous series contained elements with more atomic weight like (Te) placed before the element with less atomic weight like I.
Question 6.
How are the elements arranged Into groups and periods In the Modern Periodic Table ? Elements in a group possess similar properties, but elements in a period do not show similarities in their properties. Why?
Answer:
1. Groups and sub-groups: There are 8 vertical columns called groups, represented by Roman numerals I to VIII. Each group is divided into two sub-groups A and B. The elements present In a group and Its sub-group have similar properties.
2. Periods: The horizontal rows in Mendeleeff’s table are called ‘periods’. There are 7 periods, denoted by Arab numerals 1 to 7. A period comprises the entire range of elements after which properties repeat themselves.
3. Physical and chemical properties of elements are related to their electronic configurations, particularly the outer shell configurations.
4. The atoms of the elements in a group possess similar electronic configurations. Therefore, we expect all the elements in a group should have similar chemical properties and there should be a regular gradation in their physical properties from top to bottom.
5. SimIlarly in a period from left to right, elements get an increase In the atomic number by one unit between any two successive elements.
6. Therefore, the electronic configuration of valence shell of any two elements in a given period is not same. Due to this reason, elements along a period possess different chemical properties with regular gradation in their physical properties from left to right.
Question 7.
Explain any four factors which influence the electron affinity (Electron Gain Enthalpy)
Answer:
All the factors which influence the ionization energy would also influence the electron gain enthalpy.
- Nuclear charge: More the nuclear charge, more is the electron affinity.
- Screening effect or shielding effect: More the shells with electrons between the nucleus and the valence shell, they act as screens and decrease nuclear attractIon over valence electrons. This is called the screening effect. More the screening effect less is the electron affinity.
- PenetratIon power of the orbitals: Orbitais belonging to the same main shell have different penetration power towards the nucleus. For example : 4s>4p>4d>4f in the penetration. Therefore it is easier to add electrons in 4f than In 4s.
- Stable configuration: It is difficult to add an electron to atoms which have stable half-filled or completely filled orbitals. So, 7N(1s2,2s2,2p3) has more electron affinity value than 10(1s2,2s2,2p4) due to its half-filled configuration.
- Atomic Radius: More is the atomic radius, less is the electron affinity.
Question 8.
Define ionization energy. Explain on which factor the ionization energy depends on.
Answer:
Ionization energy: The energy required to remove an electron from the outermost orbit or shell of a neutral gaseous atom is called ionization energy.
The ionization energy of an element depends on its
1. Nuclear charge: More the nuclear charge, more is the ionization energy.
Eg: Between, Na and 7d, the chlorine atom has more ionization energy.
2. Screening effect or shielding effect: The shells with electrons between nucleus and the valence shell, act as screens and decrease nuclear attraction over valence electrons called i.e screening effect. More the screening effect, less is the Ionization energy. Eg: Between 3Li and 55Cs the element 55Cs with more inner shells has less ionization energy.
3. PenetratIon power of the orbitals: Orbitais belonging to the same main shell have different piercing power towards the nucleus.
Ex : 4s > 4p > 4d > 4f> In the penetration.
It is easier to get 4f electrons than 4s. Between 4Be (1s22s2) and 5B (1s2,2s2,2p), the element 5B has less ionization energy due to less penetration power of 2p compared to 2s.
4. Stable configuration: It is easier to remove one electron from,80 (1s2,2s2,2p4) than 7N (1s2 2s2,2p3). This is because 7N has stable half-filled configuration.
5. Atomic radius: As atomic radius Increases, the ionization energy decreases. Cg: I.E. of F is greater than that of I. because the atomic radius of I is more than that of F.
![]()
Question 9.
The atomic number of elements P, Q R, S, and T are given below :
| element | P | Q | R | S | T |
| Atomic number | 7 | 10 | 12 | 4 | 19 |
From the above table, answer the following questions:
a) Which element among these is a non-metal?
b) Which element belongs to 3rd period of periodic table?
c) Which Is an inert gas?
d) Which two elements are chemically similar?
Answer:
The electronic configuration of the elements are as follows:
Element j P Q R S T
Atomic number 17(2,5) 10(2,8) 12(2,8,2) 4(2,2) 19(2,8,8,1)
a) Element P is a non-metal since it has five valence electrons.
b) Element R belongs to third period since it has three shells.
c) Element Q is an inert gas element since It has complete octet.
d) Elements R and S are chemically similar since they have same number of valence electrons.
Question 10.
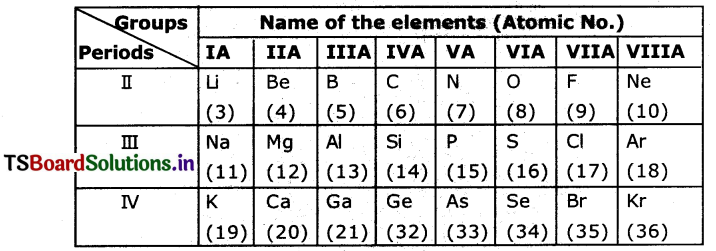
Answer the following from the above information. (March 2018)(AS3)
a) Which element possesses the higher atomic radius In the above table?
Answer:
The element with higher atomic radius is ‘K
b) Mention two pairs of & elements which forms ionic bond?
Answer:
Na, CI,; Mg, CI, etc.
c) Name the two elements having valency 2?
Answer:
Elements having valency 2 are Be, Mg, Ca, O. S, Se, etc.
d) Which element has electronic configuration of 1s22s22p4?
Answer:
Oxygen.
Do You Know
Question 1.
Are you familiar with musical notes?
Answer:
In the Indian system of music, there are seven musical notes in a scale – sa, re ga, ma, pa, da, nL In the west, they use the notations – do, re, mi, fa, so, la, ti. Naturally, there must be some repetition of notes. Every eighth note is similar to the first one and it is the fIrst note of the next scale.
Question 2.
Do you know what Mendeleeff said about the melting point of eka Al?
Answer:
‘If I hold it in mj hand, ¡t will melt’. The melting point of Ga is 30.2°C and our body temperature 37°C.
3. At the time when Mendeleeff Introduced his periodic table even electrons were not discovered. Even then the periodic table was able to provide a scientific base for the study of chemistry of elements. In his horour the With element was named Mendelevium. (Pg1s
Question 3.
Do you know how are the names of certain families of periodic table derived?
Answer:
Alkali metal family: aliquili = plant ashes, Na, K, etc…. were obtained from plant ash, group IA elements are called alkali metals family.
Chalcogen family: chalcogens = ore product, as the elements in group 16(VIA) form ores with metals. They are called as dialcogenous family.
Halogen family: halos sea salt, genus = produced. As most of the elements In group 17(VI1A) are obtained from nature as sea salt. They are called as halogen family.
Noble gases: as the elements of group 18(VIII-A) are chemically least active. They are called as noble gases. Their outer shell electronic configurations are basis for octet rule.
5. ‘Ide’ means ‘heir’ and it is used generally for a change like CIto Cl-. ‘Cl’ is chlorine atom and Cl- is chloride ion. ‘Oid’ means ‘the same’.
![]()
Some scientists suggest lanthanoids as 57La to 70Yb, some suggest them as 58Ce to 71Lu and some take 57La to 71Lu (15 elements). There is another argument that even 21Sc and 39Y should be included In lanthanoids. All these suggestions have substance because 21Sc, 39Y and 57La to 71Lu all have the similar outer shells configurations.
Also in the case of actinoids. There are different arguments like actinoids are from 90Th to 103Lr or 89Ac to 102No or 89Ac to 103Lr. (Pg135)