Telangana TSBIE TS Inter 1st Year Chemistry Study Material 10th Lesson p-Block Elements: Group 13 Textbook Questions and Answers.
TS Inter 1st Year Chemistry Study Material 10th Lesson p-Block Elements: Group 13
Very Short Answer Type Questions
Question 1.
Discuss the pattern of variation in the oxidation states of Boron to Thallium.
Answer:
The elements of boron family exhibit two oxidation states +1 and +3. From boron to thallium the stability of +1 oxidation state increases while the stability of +3 oxidation state decreases. This is due to inert pair effect.
Question 2.
How do you explain higher stability of Tl Cl3?
Answer:
Chlorine is good oxidising agent and Cl– ion is weak reducing agent. So chlorine can oxidise thallium to higher oxidation state i.e., to +3 and form TlCl3. As Cl– ion cannot reduce Tl3+, TlCl3 is stable.
Question 3.
Why does BF3 behave as a Lewis acid?
Answer:
BF3 is an electron deficient compound as there is no octet around boron. So it can accept a pair of electrons to get octet. Thus it behaves as a Lewis acid.
H3N → BF3
Question 4.
Is boric acid a protic acid? Explain.
Answer:
Boric acid is not protic acid but it is a Lewis acid. It accepts electrons from OH” ion of water and releases proton.
B(OH)3 + H2O → [B(OH)4]– + H+
Question 5.
What happens when boric acid is heated?
Answer:
On heating orthoboric acid above 370K forms metaboric acid HB02 which on further heating yields boric oxide B2O3
![]()
![]()
Question 6.
Describe the shapes of BF3 and BH4. Assign the hybridization of boron in these species.
Answer:
- Shape of BF3 molecule is Trigonal planar. Hybridization of ‘B’ in BF3 is sp².
- Shape of BH–4 molecule is Tetrahedral, Hybridizaion of ‘B’ in BH–4 is sp³.
Question 7.
Explain why atomic radius of Ga is less than that of Al.
Answer:
Gallium comes immediately after the first transition series in which 3d orbital is filled with electrons. Due to the poor shielding effect of these d electrons, effective nuclear charge of gallium increases. Consequently, the atomic radius of gallium is less than that of aluminium.
Question 8.
Explain inert pair effect.
Answer:
The pair of electrons in s orbital of valence shell are reluctant to participate in bond formation. That pair of (ns²) electrons is called inert pair. Due to this inert pair the stability of +1 oxidation state among boron family elements increases. This is known as inert pair effect.
Question 9.
Write balanced equations for
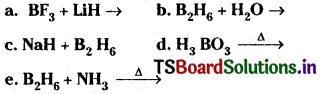
Answer:
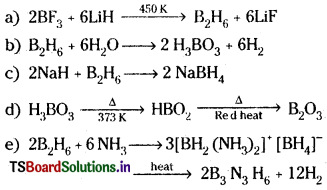
Question 10.
Why is boric acid polymeric?
Answer:
Boric acid has layer structure, in which the BO3 units are joined by hydrogen bonds. Hence, boric acid is polymeric.
Question 11.
What is the hybridization of B in diborane and borazine?
Answer:
In diborane boron is involved in sp3 hybridisation while in borazine it is in sp2 hybridisation.
Question 12.
Write the electronic configuration of group -13 elements.
Answer:
Electronic configurations:
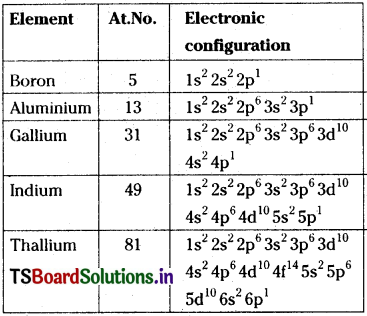
Question 13.
Give the formula of borazine. What is its common name?
Answer:
The formula of borazine is B3N3H6. It is commonly known as inorganic benzene.
Question 14.
Give the formulae of
a) Borax
b) Colemanite
Answer:
a) Formula of Borax = Na2B4O7.10H2O.
b) Formula of Colemanite = Ca2 B6 O11. 5H2O
![]()
Question 15.
Give two uses of aluminium.
Answer:
- Aluminium is used in making electrical cables.
- It is used in making trays, picture frames etc.
- Alloys of ‘Al’ are used in making parts of aircrafts, automobiles etc.
- ‘Al’ powder is used in Aluminothermite
Question 16.
What happens when
a) Li AlH4 and BCl3 mixture in dry ether is warmed and
b) orax is heated with H2SO4?
Answer:
a) Diborane is formed.
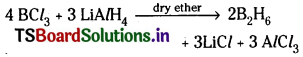
b) Boric acid is formed.
Na2B4O7 + H2SO4 + 5H2O → 4H3BO3 + Na2SO4
Question 17.
Sketch the structure of Orthoboric acid.
Answer:
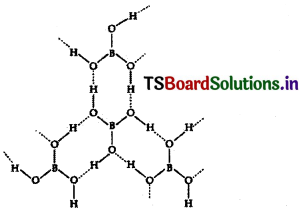
Structure of boric acid :
The dotted lines represent hydrogen bonds
Question 18.
Write the structure of AlCl3 as a dimer.
Answer:
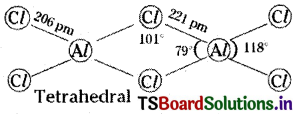
Question 19.
Metal borides (having B) are used as protective shield – why?
Answer:
10B isotope has high ability to absorb neutrons. So metal borides are used in nuclear industry as protective shields and control rods.
Short Answer Questions
Question 1.
Write reactions to justify amphoteric nature of aluminium.
Answer:
Aluminium dissolves in both acids and alkalies. Aluminium dissolves in dilute HCl and liberates dihydrogen.
2Al + 6HCl → 2 AlCl3 + 3H2.
Aluminium also dissolves in aqueous alkali and liberates dihydrogen.
2 Al + 2NaOH + 6H2O → 2 Na+ [Al(OH)4]– + 3H2
These reactions indicate the amphoteric nature of aluminium.
Question 2.
What are electron deficient compounds? Is BCl3an electron deficient species? Explain.
Answer:
The compounds in which the central atom is having less than octet electrons are called electron deficient compounds.
BCl3 is an electron deficient compound since it is not having octet around boron.
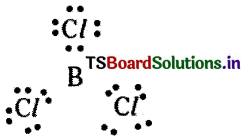
BCl3 molecule accepts an electron pair to achieve stable electronic configuration and thus behave as Lewis acid.
Eg : BCl3 molecule easily accepts alone pair of electrons from ammonia to form BCl3. NH3.
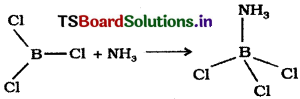
Question 3.
Suggest reasons why the B-F bond lengths in BF3 (130 pm) and BF–4 (143 pm) differ.
Answer:
In BF3 there is back bonding. Boron accepts a pair of electrons from fluorine to complete octet. BF3 exists in the following resonance structures due to back bonding. Due to the resonance hybridisation each B-F bond gets some double bond character. So the B-F length will be less than the sum of the covalent radii of boron and fluorine.
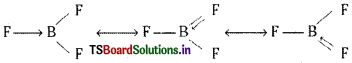
In BF–4 there is no back bonding. So there is no double bond character. This is because in BF–4 boron already had octet. Hence the B – F bond length in BF–4 is greater than in BF3.
Question 4.
B – Cl bond has a bond moment. Explain why BCl3 molecule has zero dipole moment.
Answer:
Dipolemoment of a molecule is vector sum of the dipole moments of all the bonds in the molecule. Although B – Cl bonds have bond moments, since the three bonds are oriented at an angle 120° to one another, the three bond moments give a net sum of zero as the resultant of any two is equal and opposite to the third.
So the dipole moment of BF3 is zero.
![]()
Question 5.
Explain the structure of boric acid.
Answer:
In boric acid boron is in sp² hybridisation forming planar borate units. Between the planar borate units the hydrogen atoms of the one OH group of boric acid form hydrogen bond with oxygen atom of another OH group of another boric acid unit. Thus several boric acid molecules associate through hydrogen bonds forming a layered lattice structure.
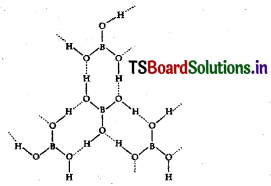
Structure of boric acid :
The dotted lines represent hydrogen bonds.
Question 6.
What happens when
a) Borax is heated strongly?
b) Boric acid is added to water?
c) Aluminium is heated with dilute NaOH?
d) BF3 is treated with ammonia?
e) Hydrated alumina is treated with aq.NaOH solution?
Answer:
a) Borax on heating strongly decomposes into NaBO2 and B2O3 forming a transparent glassy bead.
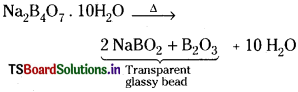
b) When boric acid is added to water it accepts a pair of electrons from OH– ion and releases proton, thus forming acidic solution.
B(OH)3 + H2O → [B (OH)4]– + H+
c) When aluminium is heated with dilute NaOH, aluminium dissolves forming tetrahydroxo aluminate (III) and liberates hydrogen gas.
2Al + 2 NaOH + 6H2O → 2 Na+ [Al (OH)4]– + 3H2
d) BF3 combines with ammonia by accepting a lone pair of electrons from ammonia.
H3N: → BF3
e) Hydrated alumina when heated with NaOH, dissolves in NaOH forming tetrahydroxo aluminate (III).
Al (OH)3 + NaOH → Na [Al(OH)4]
Question 7.
Give reasons
a) Cone. HNO3 can be transported in aluminium container.
b) A mixture of dil. NaOH and aluminium pieces is used to open drain.
c) Aluminium alloys are used to make aircraft body.
d) Aluminium utensils should not be kept in water overnight.
e) Aluminium wire is used to make transmission cables.
Answer:
a) Cone. HNO3 can be transported in aluminium container:
Concentrated nitric acid renders the aluminium passive by forming a protective oxide layer on the surface. So aluminium do not react with concentrated nitric acid. Thus the aluminium containers can be used to carry concentrated nitric acid.
b) A mixture of dil. NaOH and aluminium pieces is used to open drain:
When aluminium pieces react with dilute NaOH, dihydrogen gas will be evolved, with high pressure, which can open the drain.
c) Aluminium alloys are used to make aircraft body:
Because of light weight and hardness aluminium alloys are used to make aircraft body.
d) Aluminium utensils should not be kept in water overnight :
Aluminium does not react with pure water but when salts are present in water they remove the oxide layer on the surface making aluminium reactive. Then aluminium reacts with water slowly. So aluminium vessels should not be kept in water overnight.
e) Aluminium wire is used to make transmission cables:
Aluminium is a good conductor of electricity. So it is used in transmission cables. ‘
Question 8.
Explain why the electronegativity of Ga, In and Tl will not vary very much.
Answer:
Due to the poor screening effect of d electrons in Ga and In and due to poor screening effect of d – and f-electrons in Thallium the atomic sizes of Gallium, Indium and Thallium do not vary very much. Further the effective nuclear charge increases. So the electronegativities of Ga, In and T1 will not vary very much.
Question 9.
Explain borax bead test with a suitable example.
Answer:
Borax Bead Test:
This test is useful for the identification of coloured metal radicals in qualitative analysis. On heating, borax swells into a white, opaque mass of anhydrous sodium tetraborate. When it is fused, borax glass is obtained. This contains sodium metaborate and B2O3. The boric anhydride combines with metal oxides to form metal metaborates as coloured beads. The reactions are as follows.
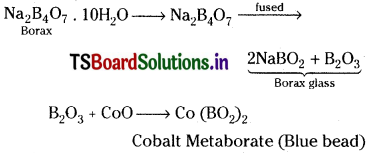
Question 10.
Explain the structure of diborane. (AP Mar. 17; AP & TS 16, ’15; IPE ’13, ’11, ’09) AP Mar. ’19; Mar. 18 (TS)]
Answer:
Structure of diborane:
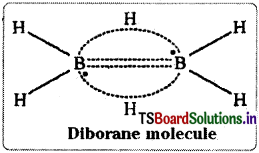
Electron diffraction studies have shown that diborane contains two coplanar BH2 groups. Its structure can be represented as
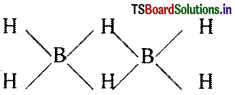
The four H atoms present in the BH2 groups are known as terminal hydrogen atoms. The remaining two H atoms are called Bridge hydrogens. The two bridge hydrogens lie in a plane perpendicular to the plane of the two BH2 groups. Out of the two bridge hydrogens, one H atom lies above the plane and the other H atom lies below the plane.
Orbital structure of Diborane :
In Diborane each boron atom undergoes sp³ hybridisation resulting in four equivalent sp³ hybrid orbitals. Three of these orbitals have one electron each and the fourth hybrid orbital is vacant. In the formation of B – H – B bridge, sp³ hybrid orbital with one electron from one boron atom, Is orbital of one bridge hydrogen and the vacant sp³ hybrid orbital of the second boron atom overlap as shown below.
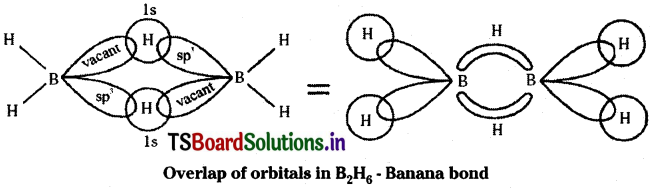
The two B – H – B bridges present in Diborane are abnormal bonds. They are considered as three centered two electron bonds. This type of bond is called banana bond or tau bond. In this type of bond a pair of electrons holds three atoms. In Diborane there are two such bridges. This Diborane structure can also be represented as shown here.
Question 11.
Explain the reactions of aluminium with acids.
Answer:
Aluminium reacts with dilute or cone. HCl liberating H2.
2 Al + 6HCl → 2 AlCl3 + 3H2
With dil. H2SO4 it reacts slowly in cold condition but reacts fast in hot condition.
2 Al + 3H2SO4 → Al2 (SO4)3 + 3H2
With cone. H2SO4 it liberates SO2.
2 Al + 6H2SO4 → Al2(SO4)3 + 6H2O + 3SO2
With very dil. HNO3 it gives NH4NO3.
8Al + 30 HNO3 → 8A/(NO3)3 + 3 NH4 NO3 + 9H2O
Concentrated nitric acid renders the aluminium passive.
![]()
Question 12.
Write a short note on the anomalous behaviour of boron in the group – 13.
Answer:
- Boron is a non-metal. ‘Al’ is amphoteric. Ga, In and Tl are metals.
- Boron always forms covalent compounds, while the others form ionic compounds.
- B2O3 is an acidic oxide. The trioxides of others are either amphoteric or basic in nature.
- B(OH)3 is an acid while the hydroxides of other elements are either amphoteric or basic.
- Simple borates and silicates can polymerise readily forming polyacids while others do not form such polymers.
Question 13.
Aluminium reacts with dil. HNO3 but not with cone. HNO3 – explain.
Answer:
Cone, nitric acid oxidises the aluminium to aluminium oxide. This forms as a layer on the surface of the metal which prevents the further reaction. This oxide layer acts as a protective layer. Thus aluminium becomes passive with cone. HNO3 and does not react with cone. HNO3.
In dilute HNO3 oxide layer on the surface will be dissolved, then the hydrogen liberated from nitric acid reduces it to ammonium nitrate.
Question 14.
Give two methods of preparation of diborane.
Answer:
1) Diborane can be prepared by treating boron trifluoride with LiAlH4 in diethyl ether.
4 BF3 + 3 LiAlH4 → 2 B2H6 + 3 LiF + 3 AlF3
2) In the laboratory it is conveniently prepared by the oxidation of sodium boro-hydride with iodine.
2 Na BH4 + I2 → B2H6 + 2NaI + H2
3) On large scale it is prepared by reaction of BF3 with sodium hydride.
![]()
Question 15.
How does diborane react with
a) H2O b) CO c) N(CH3)3?
Answer:
a) Diborane hydrolyses in water liberating hydrogen gas with the formation of boric acid.
B2H6 (g) + 6H2O → 2 B(OH)3 (aq) + 6H2(g)
b) Diborane forms an addition product with CO. In this reaction diborane undergoes symmetric cleavage.
B2H6 + 2 CO → 2 BH3. CO
c) While reacting with N(CH3)3 also diborane undergoes symmetric cleavage forming the addition compound.
B2H6 + 2 N (CH3)3 → 2 BH3 . N (CH3)3
Question 16.
Al2O3 is amphoteric – explain with suitable reactions.
Answer:
Al2O3 reacts with both acids and bases. While reacting with acids it behaves like base. In the reaction with bases it behaves like acid. So it is amphoteric.
Reaction with acid:
Al2O3 + 6HCl → 2 AlCl3 + 3H2O
Reaction with base:
Al2O3 + 2NaOH → 2Na AlO2 + H2O
Question 36.
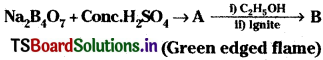
Identify A and B.
Hint: A = H3BO3; B = (C2H5)3 BO3.
Answer:
When borax is heated with cone. H2S04 boric acid is formed.
Na2B4O7 + H2SO4 → Na2SO4 + H2B4O7
H2 B4O7 + 5H2O → 4 H3BO3
Na2 B4O7 + H2SO4 + 5H2O → Na2SO4 + 4H3BO3
The boric acid forms triethyl borate when heated with cone. H2SO4 and ethyl alcohol. This burns with green edged flame.
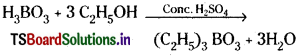
Long Answer Questions
Question 1.
How are borax and boric acid prepared? Explain the action of heat on them.
Answer:
1) Borax occurs naturally as tincol, associated with impurities in sediments of lakes. Borax is highly soluble in hot water but crystallises in cold water. So the tincol is dissolved in hot water and filtered to remove insoluble impurities. Then the filtrate is cooled to get the pure borax.
Borax on heating decomposes into sodium metaborate and boron trioxide which form a transparent glassy bead.
![]()
2) When borax is heated with cone. H2SO4, boric acid is formed.
Na2B4O7 + H2SO4 + 5H2O → Na2SO4 + 4 H3 BO3.
Boric acid on heating first gives meta-boric acid at low temperature but at red heat condition forms boron trioxide.
![]()
Question 2.
How is diborane prepared? Explain its structure.
Answer:
Preparation of Diborane :
1) Boron trifluoride on reduction with LiH at 450K gives Diborane.
2BF3 + 6 LiH → B2H6 + 6LiF
2)vOn passing silent electric discharge through a mixture of boron trichloride and hydrogen, diborane is formed.

Molecules in which the central atom gets less than octet configuration are called electron-deficient molecules.
Ex : (1) Diborane B2H6
(TrBoron trifluoride BF3.
Structure of diborane:
Electron diffraction studies have shown that diborane contains two coplanar BH2 groups. Its structure can be represented as

The four H atoms present in the BH2 groups are known as terminal hydrogen atoms. The remaining two H atoms are called Bridge hydrogens. The two bridge hydrogens lie in a plane perpendicular to the plane of the two BH2 groups. Out of the two bridge hydrogens, one H atom lies above the plane and the other H atom lies below the plane.
Orbital structure of Diborane :
In Diborane each boron atom undergoes sp³ hybridisation resulting in four equivalent sp³ hybrid orbitals. Three of these orbitals have one electron each and the fourth hybrid orbital is vacant. In the formation of B – H – B bridge, sp³ hybrid orbital with one electron from one boron atom, Is orbital of one bridge hydrogen and the vacant sp³ hybrid orbital of the second boron atom overlap as shown below.
The two B – H – B bridges present in Diborane are abnormal bonds. They are considered as three centered two electron bonds. This type of bond is called banana bond or tau bond. In this type of bond a pair of electrons holds three atoms. In Diborane there are two such bridges. This Diborane structure can also be represented as shown here.
![]()
Question 3.
Write any two methods of preparation of diborane. How does it react with
i) Carbon monoxide and ii) Ammonia?
Answer:
i) Preparation of diborane :
1) Diborane can be prepared by treating BF3 with Li A/ H4 in diethyl ether.
4 BF3 + 3 Li AlH4 → 2 B2H6 + 3 LiF + 3 AlF3
2) In the laboratory it is conveniently prepared by the oxidation of sodium borohydride with iodine.
2 Na BH4 + I2 → B2H6 + 2 Nal + H2
3) On a large scale it is prepared by the reaction of BF3 with sodium hydride.
![]()
ii) a) Diborane forms an additional product with CO with symmetric cleavage.
B2H6 + 2CO → 2BH3.CO
b) With ammonia it forms an addition product with asymmetric cleavage which on heating forms borazine which is known as inorganic benzene.
![]()