These TS 10th Class Physical Science Chapter Wise Important Questions Chapter 8 Chemical Bonding will help the students to improve their time and approach.
TS 10th Class Physical Science Important Questions Chapter 8 Chemical Bonding
1 Mark Questions
Question 1.
How is covalent bond formed?
Answer:
A covalent bond is formed by the sharthg of electrons between two atoms.
Eg: Bonding In H2 molecule, O2 molecule, N2 molecule etc.
Question 2.
What is ‘Octet rule’?
Answer:
Octet rule: The presence of 8 electrons in the outermost shell of an atom or a molecule is called ‘octet rule.
Question 3.
What is’Bondlength’?
Answer:
Bond length: ft is the inner-nucleus distance between the two atoms in a molecule. It is measured In Angstrom, 1 Å = 10-8 cm.
Question 4.
What is a chemical bond?
Answer:
An attractive force between two atoms In a molecule Is called a chemical bond.
Question 5.
How is a cation formed?
Answer:
A cation Is formed when an atom loses electrons.
![]()
Question 6.
How is an anion formed?
Answer:
An anion Is formed when an atom gains electrons.
Question 7.
What are the forces present in an Ionic bond?
Answer:
Electrostatic forces of attraction are present in ionic bonds.
Question 8.
Which type of compounds are more soluble In polar solvents?
Answer:
Ionic compounds are more soluble In polar solvents.
Question 9.
Which compounds exhibit high melting and boiling points?
Answer:
Ionic compounds exhibit high melting and boiling points.
Question 10.
What is electronic configuration’?
Answer:
A systematic arrangement of electrons In the atomic orbits Is called electronic configuration.
Question 11.
Why are molecules more stable than atoms?
Answer:
Molecules have lower energy than that of the combined atoms. Molecules are more stable than atoms since chemical species with lower energy are more stable.
Question 12.
Which forces are weaker forces and where are they operative?
Answer:
- Vander Waal’s forces are very weak forces.
- They are operative between non-polar molecules.
Question 13.
Which compounds exhibit low melting and boiling points?
Answer:
Covalent compounds exhibit low melting and boiling points when compared to ionic compounds.
Question 14.
What is ionic linkage?
Answer:
When two oppositely charged Ions are engaged in a bond, it is known as ionic linkage.
Question 15.
On which factors do anions depend?
Answer:
- Atomic size
- Ionization potential
- Electron affinity
- Electronegativity
Question 16.
What is VSEPRT’?
Answer:
VSEPRT means ‘valence – shell – electron – pair repulsion – theory’.
Question 17.
How do you know the valence of a metal?
Answer:
The number of electrons lost from a metal atom is the valence of its elements which is equal to its group number. Eq: Na and Mg have valences 1 and 2 respectively.
Question 18.
How do you know the valence of a non-metal?
Answer:
The number of electrons gained by a non-metal element for its atom to attain octet configuration is its valency, which is equal to 8 – its group number. Eg: The valency of chlorine is (8 – 7) = 1.
![]()
Question 19.
Why do atoms combine and form molecules?
Answer:
The energy of molecule is less than the total energy of constituent atoms. Therefore atoms combine and go to a stable state of lower energy.
Question 20.
What is ‘orbital concept of bond formation’?
Answer:
Atoms with hair-filled or vacant orbitals try to get paired electrons in those orbitals by bond formation i.e., by losing, gaining or sharing of electrons.
Question 21.
Name the bonds present In the molecules i) BaCl2 ii) C2H4.
Answer:
(i) In BaCl2 – Ionic.
(ii) In C2H4 – Covalent (double bond H2C = CH2)
Question 22.
What s the use of VSEPR Theory?
Answer:
It predicts shapes of molecule without consideration of hybridization.
Question 23.
Why a molecule of Hydrogen is more stable than the uncombined atoms?
Answer:
When a molecule of hydrogen is formed from the atoms, energy is released (104 Kcal/ mol) Thus the molecule possessing lower energy is more stable that the atoms.
Question 24.
Why ionic compounds are good electrolytes?
Answer:
- Electrolytes produce ions in solution, which carry current.
- Ionic compounds is the fused state and aqueous solutions contain ions moving freely. Hence they conduct electricity.
Question 25.
When is ionic bond termed between atoms?
Answer:
Ionic bond is readily formed between atoms of elements with a low ionisation energy and atoms of elements with high electronegativity.
Question 26.
What is crystal lattice’?
Answer:
In a crystal of an ionic compound each Ion Is surrounded by oppositely charged ions Th ions arrange themselves at an optimum distance with regular periodicity in a well-defined three-dimensional network called crystal lattice.
Question 27.
What is lattice energy?
Answer:
the energy released when gaseous positive and negative ions are brought together from infinity to form one mole ionic crystal Is called lattice energy.
Question 28.
Represent the formation of nitrogen molecules by Lewis notation. (AS1)
Answer:
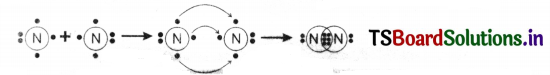
Question 29.
Represent Fluorine a molecule by using lewies notation.
Answer:

Question 30.
How many sigma and pl bonds are present in acetylene molecule?
Answer:
HC = CH, one sigma and two pi bonds are present.
Question 31.
How is a σ (sigma) bond formed?
Answer:
By the axial or tendon overlap of pure orbitals or hybrid orbitals of two atoms.
Question 32.
How is a π (pi) bond formed?
Answer:
By the lateral or parallel overlap of only Pure atomic orbitals after the σ bond formation.
![]()
Question 33.
Which type of atoms easily enter into ionic bonding?
Answer:
An atom with low ionization potential and another with high electron affinity.
Question 34.
What is a polar bond?
Answer:
A covalent bond in a heteroatomic molecule. Eg : HCl.
Question 35.
What is meant by inter-nuclear axis?
Answer:
The hypothetical line joining the center of nuclei of two atoms in a molecule.
Question 36.
What are multiple bonds?
Answer:
Double and triple bonds are multiple bonds.
Question 37.
How many σ and π bonds are in O2 molecule?
Answer:
One σ and two π bonds.
Question 38.
What are Lewis structures?
Answer:
The symbol of the atom gives the core (or kernel) of the atom is, which valence electrons are shown as dots (.), cIrcles (O) or crosses (x). Thus in Nax, Na represents the core of sodium atom and the cross (x) represents the valence electron of sodium.
Question 39.
In case of Ionic substances, a more appropriate term Is formula weight, rather than ‘molecular weight’. Why?
Answer:
Molecules are not present in ionic substances. Only ions are present. The formula of an ionic substance represents the simpler ratio of ions in one mole of crystal Hence a more appropriate term Is ‘formula weight’.
Question 40.
NaCl dissolves in water but not In benzene. Explain.
Answer:
NaCl dissolves in water because of hydration. Water being a polar molecule has positive and negative ends which hydrate Na+ and Cl– ions. Benzene being nonpolar cannot dissolve the ions of NaCl.
![]()
Question 41.
What are the bond angles in H2O and NH3 molecule?
Answer:
Bond angle In H2O molecule is 104° 3′.
Bond angle In NH3 molecule is 107°.
Question 42.
Why noble gases (inert gases) are stable?
Answer:
- Noble gases are stable because their outermost orbit contains 8 or 2 electrons.
- Noble gases exist as individual atoms.
Question 43.
What is an ion?
Answer:
An ion is an electrically charged atom (or group of atoms).
Question 44.
Sulphur dioxide is a diamagnetic molecule. Explain.
Answer:
Sulphur dioxide (SO2) is a diamagnetic molecule because it has all electrons paired (no free electrons).
Question 45.
What are the structures of sodium chloride and calcium chloride crystals?
Answer:
Sodium chloride has face-centered cubic structure. Calcium chloride has body-centered cubic structure.
Question 46.
Write a short note on bond angles.
Answer:
It is the average angle between two adjacent atoms bonded to the central atom in a molecule. Molecules with larger bond angles are more stable than those with smaller angles.
Question 47.
What Is ionisatIon? Give one example.
Answer:
- The process of removal of electron (s) from an atom or molecule is termed
Ionisation. g : Na → Na+ + e– - Dissociation of an ionic solid into constituent ions upon Its dissolution in a suitable solvent is also called ionisation. Eg : NaCl?(aq) → Na+(aq) +Cl–(aq)
Question 48.
What is a Coordination number? (ASI)
Answer:
The number of ions of opposite charge that surround a given ion of given charge is known as coordination number of that given Ion.
Question 49.
Show the formation of HCl molecule with Lewis dot structures using the information given below.
Answer:
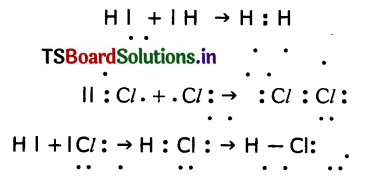
Question 50.
Bond angle of Water reduced to 104°27’ from 109.28 said, Rajesh. Is he corred? Justify your answer.
(or) Explain, why bonding angle (HOH) In water Is 104°31’ instead of 109° 281?
Answer:
Yes. He Is correct.
justification:
- In water molecule oxygen atom has two lone pairs of electrons (: 0 🙂 and two bond pairs (O- H) of electrons.
- Due to this two lone pairs of electrons shows repulsion on bond pairs.
- So, the bond angle reduced to 104°.27 from 109°,28

2 Marks Questions
Question 1.
Explain Ionic bond with suitable example.
Answer:
- Sodium (Na) looses one electron and forms Sodium ion (Na+).
Na → Na++1e– - Chlorine (Cl) gains one electron and forms chloride ion (Ci).
Cl+le– → Cl– - Positive sodium on (Na) and negative chloride (Ci) ion come together due to electrostatic forces, participate in ionic bond and form NaCl.
Na+ + Cl– → NaCl
Question 2.
Distinguish between a sigma bond and a pi bond.
Answer:
| Sigma bond | Pi bond |
| 1. It is formed by the end-on-end overlap of orbitals. | 1. It is formed by the lateral overlap of orbitals. |
| 2. It has Independent existence. | 2. It has no independent existence. |
| 3. It Is a strong bond. Because axial overlap is more. | 3. It is a weak bond. because lateral overlap is less. |
| 4. There can be only one sigma bond. | 4. There can be one or two it bonds between two atoms. |
Question 3.
Write about ‘Hydrogen bond’.
Answer:
1. Hydrogen bond Is formed between molecules in which hydrogen atom s attached to an atom of an element with large electronegativity and very small size (F. O. N). Because of hydrogen bond the molecules associate themselves and hence possess higher B.P’s and M.P’s. ,
2. The hydrogen bond formed between two molecules is called inter-molecular hydrogen bond
3. The hydrogen bond formed between different groups of the same Molecule is called intra-molecular hydrogen bond. “
![]()
Question 4.
Bring out the difference between Ionic and covalent bonds.
Answer:
| Ionic bond | Covalent bond |
| 1. It is formed by transference of electrons from one atom to the other. | 1. It is formed by the sharing of electron pairs by two atoms. |
| 2. Electrostatic. | 2. Not electrostatic, but rigid. |
| 3. Ionic substances are formed by ionic bonds. | 3. Molecules are formed by covalent bond. |
| 4. on – directional. | 4. Directional. |
Question 5.
Write about VSEPR theory.
Answer:
It predicts the possible shapes of the molecules based on the number of electron pairs In the valence shell of the central atom.
| Electron pairs | Shape of molecules |
| Two | Linear |
| Three | Equilateral triangle |
| Four | Tetrahedral |
| Five | Trigonal bipyramidal |
| Six | Octahedral |
Question 6.
Write a short note on octet rule with example.
Answer:
Atoms try to get 8 electrons in their outermost shell (inert gas structure) by combining with other atoms. In order to get 8 outer electrons, atoms may lose to, gain from or share electrons with other atoms.
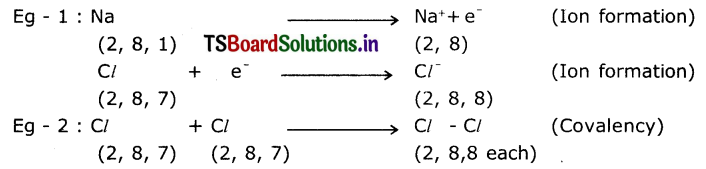
Question 7.
Why a large amount of energy is needed to remove an electron from a neutral gaseous neon atom than the energy needed to remove an electron from gaseous sodium atom?
Answer:
Na(g) + 118.4 K.cal → Na’ + e
Ne(g) + 497.0 K.cal → Ne + e
Orbitals which are fully filled are very stable, so large amount of energy is needed to remove an electron from them.
Question 8.
Why there is absorption of energy in certain chemical reactions and release of energy In other reactions?
Answer:
If bond dissociation energy of reactants is more than bond energy of products then energy ¡s absorbed n the chemical reaction.
If bond dissociation energy of reactants is less than bond energy of products then energy s released’ in the chemical reaction.
Question 9.
Electronic configuration of x is 2, 8, 1 and electronic configuration of Y is 2, 8, 7 ExplaIn what type of bond Is formed between them.
Answer:
The element with electronic configurations 2, 8, 1 is sodium which Is a metal. Similarly, the element with electronic configurations 2, 8, and 7 Is chlorine which is a non-metal. The electronegativity difference between these two elements Is more than 1.9 so they form ionic bond.
Question 10.
Why ionic compounds dissolve in polar solution and covalent compounds dissolve in non-polar solution?
Answer:
Ionic compounds are polar in nature so they are soluble in polar solvents whereas covalent compounds are non-polar in nature so they are soluble in non-polar solvents.
Question 11.
What are favourable conditions for the formation of anion?
Answer:
The favourable conditions for the formation of anion is
- High Electron Affinity.
- Low charge on the anion.
- Small size of the non-metal.
- Configuration is very near to inert gas configuration.
Question 12.
Bond angle of ammonia reduced to 107°48 from 109°28” said Ramya. Is she correct? Justify your answer.
Answer:
Yes. She Is correct.
Justification:
- In NH3 there are three bond pairs (N-H) and one lone pair of electrons (N) Is present around the central atom of nitrogen.
- So, the lone pair electron shows repulsion on bond pairs.
- Hence, to minimise the repulsion force bond angle changes from 109°28 to 107°48.
- At the same time the shape also changes from tetrahedral to pyramidal.

Question 13.
Write the ‘Octet Rule’. How does Mg (12) get stability while reacting with chlorine as per this rule?
Answer:
Octet rule: The atoms of elements tend to undergo chemical changes that help to leave their atoms with eight outer-shell electrons.
Electronic configuration of ‘Mg’ is – 2, 8, 2
So, it loses two electrons in its outer shell to get octet configuration and becomes Mg+2.
So, it gives 2 electrons to two chlorine atoms to form Magnesium chloride (Mgcl2) and gets stability.
![]()
Question 14.
Draw the diagram to show the formation of Oxygen molecules by Valence bond theory.
Answer:

4 Marks Questions
Question 1.
If the electronic configurations of atoms A and B are 3S2, 2S2, 2P6,3S2, 3P1’ and 2, 61S2, 2P4 respectively, then
a) Which atom forms negative ion
b) Which atom forms positive ion?
c) What Is the valency of atom A?
d) What Is the molecular formula of the compound form by atoms A and B?
Answer:
Given electronic configuration of atom A is Is’ 2s2 2p6 3s2 3p1 i.e., Aluminium and B is 1S2, 2S2, 2p6 i.e., Carbon.
a) The atom ‘B’ tends to form negative ion by gaining two electrons order to get nearest inert gas neon configuration is 1s22s22p6.
b) The atom ‘A’ tends to form positive ion by losing three electrons n order to get nearest inert gas. Neon configuration is 1s22s22p6.
c) Valency of atom ‘A’ is ‘3’.
d) According to Criss-Corss method, the molecular formula of the compound formed by atoms both A and B Is A2B3 i.e., AlO3.
Question 2.
a) Draw the electron dot structures of Sodium, Oxygen and Magnesium.
b) Show in the form of a picture, the formation of Na20 and MgO.(AS5)
Answer:
a) Sodium atom has one valency electron in its valency shell, so its electron dot structure Is Nax(or)Na Oxygen atom has two valency electrons in its valency shell, so Its electron dot structure is O (or) O:
Magnesium atom also having two valency electrons in its valency shell, so its electron dot structure is Mg: (or) Mg :
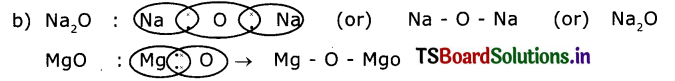
Question 3.
Explain VSEPRT theory.
Answer:
VSEPRT theory:
The full form of VSEPRT is Valence Shell Electron Pair Repulsion Theory.
- VSEPRT proposes that molecules get specific shapes due to repulsion between bond pair and lone pair.
- Lone pair occupies more space around the central atom than bond pair.
- Thus the presence of lone pairs on the central atom causes slight distortion of bond angles from the expected regular shape.
- If two bond pairs without lone pair then shape of the molecule is linear with bond angle 180°.
- It three bond pairs without lone pair then shape is trigonal planar with bond angle 120°.
- If there are four bond pairs without lone pair then shape is tetrahedron with bond angle 109°28.
- If there are three bond pairs and one lone pair then due to repulsions between bond pair and lone pair the shape is pyramidal.
- If there are two bond pairs and two lone pairs then due to lone pair and bond pair repulsions the shape of the molecule is V.
Question 4.
Explain the formation of BF3 molecule with the help of Valency Bond theory.
Answer:
Formation of BF3 molecule:
- 3B has electronic configuration 1s22s22px1.
- The excited electronic configuration of 5B is 1s2 2s1 2px1 2p1y.
- As It forms three identical B-F bonds In BF3.
- It Is suggested that excited ‘B atom undergoes hybridisation.
- There is an intermixing of 2s, 2px, 2py orbitals and their redistribution into three Identical orbitals called sp2 hybrid orbitals.
- For three sp2 orbitals to get separated to have minimum repulsion the angle between any two orbitals is 120° at the central atom and each sp2 hybrid orbitals.

- Now three fluorine atoms overlap their 2p2 orbitals containing unpaired electrons (F9, 1s2 2s2 2px2 2px2 2py2,2pz1) the three sp2 orbitals of ‘B that contain unpaired electrons to form three σsp2-p bonds.
Question 5.
What Is hybridisatlon?
Answer:
In the formation of molecules, the atomic orbitals of the atoms may hybridise.
- It is the process of mixing up of atomic orbitals of an atom to form identical hybrid orbitals.
- This takes place only during the formation of bond.
- There should not be much difference in the energies of the orbitals that hybridise.
- The number of hybrid orbitals formed is equal to the number of hybridising atomic orbitals.
- Hybrid orbitals form sigma bonds only not t bonds.
Question 6.
What are the important characteristic features of hybridisation?
Answer:
- Orbitals on a single atom only would undergo hybridization.
- The orbitals involved In hybridisation should not differ largely In their energies.
- The number of hybrid orbitals formed is equal to the number of hybridising orbitals.
- The hybrid orbitals form stronger directional bonds than the pure s, p, d atomic orbitals.
- It is the orbitals that undergo hybridisation and not the electrons.
- Concept of hybridisation is useful in explaining the shape of molecules.
Question 7.
Can you suggest an experiment to prove that ionic compounds possess strong bonds when compared to that of covalent bonds? Explain the procedure.
Answer:
- Take a small amount of sodium chloride (NaO) on a metal spatula (having an insulated handle).
- Heat it directly over the flame of a burner.
- We will see that sodium chloride (NaCl) does not melt easily.
- Sodium chloride melts (and becomes a liquid) only on strong heating.
- This shows that sodium chloride which is an Ionic compound possesses strong bonds.
- So it has a high melting point.
- Whereas covalent compounds like naphthalene and carbon tetra chloride has low boiling points such as 80° C and 77° C respectively.
- The force of attraction between the molecules of a covalent compound is very weak.
- Only a small amount of heat energy Is required to break these weak molecular forces, due to which covalent compounds have low melting points and low boiling points.
Question 8.
A list of five substances is given.
a) NaCl
B) CH4
C) CCl4
d) N2
e) Ar
Which of these substances correspond to the options (i) to (x)
i) A substance with high melting point
ii) A substance with low heat of vapourisation
iii) Diatomic
iv) An insulator
v) Soluble In water
vi) A liquid which is immutable in water
vii) Monoatomic
viii) A gas which Is insoluble In water
ix) An Ionic compound
x) Covalent compound
Answer:
(i) NaCl
(ii) CCl4
(iii) N2
(iv) CCl4
(v) NaCl
(vi) CCl4
(vii) Ar
(viii) N2 and CH4
(ix) Nacl
(x) CH4, CCl4, N2.
Question 9.
Explain the formation of Ammonia molecules by using Lewis notation.
Answer:
Ammonia (NH3) molecule: In ammonia molecule, three N – H single covalent bonds are present -Electron configuration of 7N is 2, 5 and 1H is 1. Nitrogen (N) needs 3 electrons to have octet In Its valence shell. Therefore it shows ‘3’ electrons with ‘3’ hydrogen(H) atoms to form ammonia (NH3) molecule.
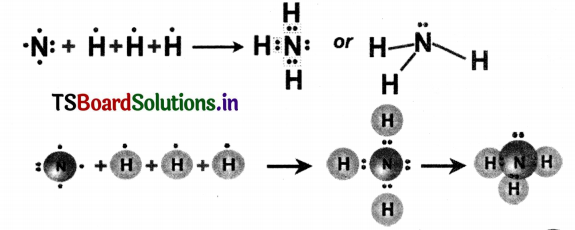
Question 10.
Explain the formation of Methane molecules by using Lewis notation.
Answer:
Methane (CH4) molecule: In the formation of methane, (CH4) molecule, carbon contributes 4 electrons (one electron to each hydrogen atom) and 4 hydrogen atoms contribute one electron each. Thus in CH4 molecule, there are four CH4 covalent bonds as shown below:
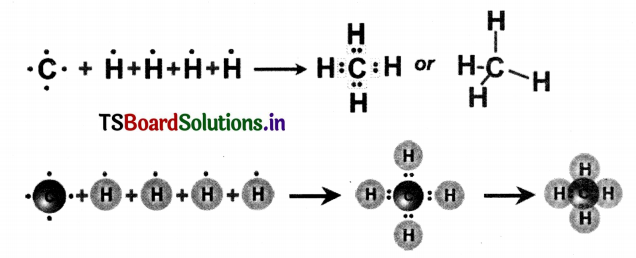
![]()
Question 11.
Write the difference between ionic bond and covalent bond?
Answer:
| Ionic bond | Covalent bond |
| 1. Ionic bond is formed by the transfer of one or more electrons. | 1. Covalent bond is formed sharing of electrons. |
| 2. It is formed between metal and non-metal. | 2. It is formed between non-metals. |
| 3. It is also called electrovalent bond and Is due to electro valency. | 3. It Is called electron pair bond and is due to covalency. |
| 4. Ionic bond consists of electrostatic force of attraction between the oppositely charged eons. | 4. Covalent bond consists of shared pair or pairs of electrons which an attracted by both the nuclei. |
| 5. Ionic bond is non rigid and non-directional. | 5. Covalent bond is rigid and directional. |
| 6. lonic bond is polar in nature. | 6. Covalent bond may be polar or non-polar. |
Question 12.
An element X has 3 electrons In the outermost shell of the atom. (AS1)
a) State whether ‘X’ Is a metal or a non-metal?
b) Write the symbol of Its ion?
c) Write the formula of its oxide?
Answer:
a) An element which has 3 electrons In the outermost shell is Aluminium. It is confirmed by writing electronic configuration 1S2 2S22P63S13p1.
b) The symbol is Al+3. It is a metal
c) Its oxide Is Al2O3
4Al + 3O2 → 2Al2O3
Question 13.
Give electron dot formula for the following.
a) Magnesium chloride
b) Carbon-di-oxide
c) Carbon tetrachloride
d) Hydrogen bromide
Answer:
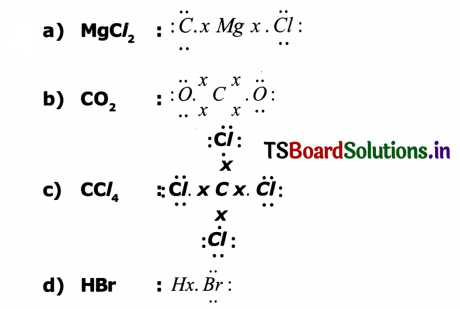
Do You Known
Davy’s experiment
Humphry Davy (1778-1829), a Professor of Chemistry at the Royal Institute in London, constructed a battery of over 250 metallic plates. In 1807, using electricity from this battery, he was able to extract highly reactive metals like potassium and sodium by electrolysis of fused salts (Page 151)

It was seen that the metal part of the compound migrated towards the negative electrode and the
non-metal part towards the positive electrode. So it was proposed that metals are responsible for
positively charged particles and non-metals are responsible for negatively charged particles. The oppositely charged particles are held together by electrostatic forces in a compound. Do you agree with this explanation? Why? While this explanation could explain bonding in Nacl, KCl etc. It could not explain bonding in carbon compounds or diatomic molecules of elements.

2. The number of electrons lost from a metal atom is the valence of its element which is equal to its group number (Page 157) Eg: Na and Mg have valencies 1 and 2 respectively
3. The number of electrons gained by a non-metal element for its atom is it valency, which is equal to (8 – its group number) (Page 157) Eg: The valency of chlorine is (8-7) – 1
4. An angstrom (Å) is a unit of length equal to 10-10 m, or 0.1 nanometre, or 101 picometres. 1 nanometre = 10-9 metre; 1 picometre = 10-12 metre. (Page 164)