Telangana TSBIE TS Inter 1st Year Chemistry Study Material 11th Lesson p-Block Elements: Group 14 Textbook Questions and Answers.
TS Inter 1st Year Chemistry Study Material 11th Lesson p-Block Elements: Group 14
Very Short Answer Type Questions
Question 1.
Discuss the variation of oxidation states in the group-14 elements.
Answer:
The elements of group-14 exhibit two oxidation states +2 and +4. The stability of +2 oxidation state increases from carbon to lead while the stability of +4 oxidation state decreases from carbon to lead. Carbon also exhibits different oxidation states from -4 to +4.
Question 2.
How the following compounds behave with water?
a) BCl3 b) CCl4
Answer:
a) BCl3 hydrolyses in water
BCl3 + 3H2O → H3BO3 + 3HCl
b) CCl4 do not hydrolyse in water; and in-soluble in water forming separate layer.
Question 3.
Are BCl3 and SiCl4 electron deficient compounds? Explain.
Answer:
BCl3 is an electron deficient molecule since boron does not have octet. It can accept an electron pair Lewis bases and act as Lewis acid.
SiCl4 is not an electron deficient molecule. Silicon has octet in this compound.
Question 4.
Give the hybridization of carbon in
a. CO2-3 b. diamond c. graphite d. fullerene [AP. TS ’16; IPE ’14 Mar. ’18 (TS)]
Answer:
Hybridization of carbon in
a. CO2-3 – sp² hybridisation
b. diamond – sp³ hybridisation
c. graphite – sp² hybridisation
d. fullerene – sp² hybridisation
Question 5.
Why is carbon monoxide poisonous? [Mar. 18 (AP) (AP, TS 16; Mar. 13)]
Answer:
‘CO’ gas is highly poisonous because it has the ability to form a stable complex with haemoglobin.
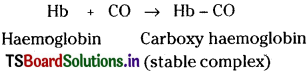
Carboxy haemoglobin is 300 times more stable than oxyhaemoglobin.
![]()
Question 6.
What is allotropy? Give the crystalline allotropes of carbon.
Answer:
The existence of an element in different physical forms but possessing similar chemical properties is known as allotropy.
The three crystalline allotropes of carbon are diamond, graphite and fullerenes.
Question 7.
Classify the following oxides as neutral, acidic, basic or amphoteric.
a. CO b.B2O3 c. SiO2 d. CO2 e. Al2O3 f. PbO2 g. Tl2O3
Answer:
a. CO – neutral
b.B2O3 – acidic
c. SiO2 – acidic
d. CO2 – acidic
e. Al2O3 – amphoteric
f. PbO2 – amphoteric
g. Tl2O3 – basic
Question 8.
Name any two man-made silicates. [AP ’15; IPE ’14]
Answer:
- Glass
- Cement
Question 9.
Write the outer electron configuration of group -14 elements.
Answer:
The elements of group 14 are Carbon (C); Silicon (Si); Germanium (Ge); Tin (Sn) and Lead (Pb).
Electronic configurations:
Carbon – 1s²2s²2p²
Silicon – 1s² 2s²2p63s²3p²
Germanium – 1s²2s²2p63s²3p63d104s²4p²
Tin – 1s² 2s² 2p63s² 3p63d104s²4p6 4d105s²5p²
Lead – 1s²2s²2p63s²3p63d104s²4p64d10 4f14 5s²5p65d10 6s² 6p²
The general outer electronic configuration of group 14 elements is ns²np².
Question 10.
How does graphite function as a lubricant? [TS Mar. 19; (TS ’ 15; Mar. 11)]
Answer:
Graphite has two – dimensional layer structure and these layers can slide easily one over the other. Hence graphite is used as a lubricant.
Question 11.
Graphite is a good conductor – explain. [AP Mar. ’17, ’15 ’09]
Answer:
Graphite has free p-electrons available. These electrons freely move throughout the crystallattice. Hence graphite acts as a good conductor.
(0r)
In graphite each carbon atom is in sp² hybridised state.
![]()
Question 12.
Explain the structure of silica.
Answer:
Silica is gaint polymeric molecule. Every silicon in silica is in sp³ hybridisation. It is surrounded by 4 oxygen atoms tetrahedrally. Every oxygen is in bond with two silicon atoms.

Question 13.
What is ‘synthesis gas’?
Answer:
A mixture of CO and H2 (water gas) is called synthesis gas. Because it is used in the synthesis of compounds such as methyl alcohol. It is obtained by passing steam over red hot coke.
C + H2O → CO + H2
Question 14.
What is ‘producer gas’ [TS ’15]
Answer:
A mixture of CO and N2 obtained by passing air over red hot coke is called producer gas.
![]()
Question 15.
Diamond has high melting point-explain.
Answer:
Diamond is a three-dimensional giant molecule. The valency electrons of each carbon take part in bonding. The C – C bonds in it are very strong. Hence diamond has a high melting point (MP).
Question 16.
Give the use of CO2 in photosynthesis. [IPE ’14]
Answer:
Plants absorb CO2 from air, water from soil and convert them into carbohydrates in the presence of sunlight and chlorophyll.
![]()
This is known as photosynthesis in which CO2 changes carbohydrates to glucose.
Question 17.
How does CO2 increase the greenhouse effect?
Answer:
The increase in combustion of fossil fuels and decomposition of lime stone, etc. increases the CO2 content in the atmosphere. This may lead to increase in the greenhouse effect, thus raise the temperature of the atmosphere which might have serious consequences.
Question 18.
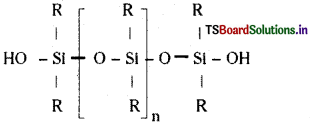
What are silicones? [AP ’15]
Answer:
Silicones:
Silicones are organo-silicon compounds. In silicones, silicon is strongly linked to oxygen and carbon. Silicones are polymers. They are formed by the hydrolysis and condensation of chlorosilanes. They bear the following structure.
Question 19.
Give the uses of silicones.
Answer:
Uses of silicones :
- Used in the preparation of silicone rubbers.
- Used in the preparation of water-proof clothes and papers.
- Used in the preparation of grease, lubricants in aeroplanes.
- Used in electrical motors as insulators.
- Used in paints and enamels.
![]()
Question 20.
What is effect of water on tin?
Answer:
Tin decomposes steam to form dioxide and dihydrogen gas.
![]()
Question 21.
Write an account of SiCl4.
Answer:
Silicon tetra chloride can be prepared by the action of chlorine on silicon.
Si + 2Cl2 → SiCl4
It is a volatile fuming liquid. It hydrolyses in water by accepting lone pair of electrons from water molecules in d – orbitals of Si, finally leading to the formation of Si(OH)4.
Uses:
- SiCl4 and NH3 mixture is used to produce smokescreens.
- SiO2 prepared from SiCl4 is used in epoxy paints, resins, etc.
Question 22.
SiO2 is a solid while CO2 is a gas – explain.
Answer:
–O — C ≡ O+ ↔ O = C = O ↔ +O ≡ C — O–
CO2 molecule is linear, carbon dioxide molecules are held together only by weak van der Waal’s forces and hence it exists as a gas at ordinary temperature.
Silicon has a three-dimensional infinite structure, in which silicon atom is tetrahedrally bonded to two silicon atoms by covalent bonds. The whole crystal of silica thus exists as a giant molecule and hence silica is a high melting solid.
Question 23.
Write the use of ZSM-5. [TS Mar. ’19]
Answer:
ZSM-5 is a zeolite used as a catalyst in petro chemical industries to convert alcohols directly into gasoline.
Question 24.
What is the use of dry ice? [AP ’15]
Answer:
Solid CO2 is called “Dry ice”. It is used as a refrigerant for ice cream and frozen food.
![]()
Question 25.
How is water gas prepared?
Answer:
When steam is passed over hot coke a mixture of CO and H2 is produced. This is known as water gas.
C + H2O → CO + H2
Question 26.
How is producer gas prepared?
Answer:
When air is passed over hot coke a mixture of CO and N2 is produced. This mixture of CO and N2 is known as producer gas.
![]()
Question 27.
C – C bond length in graphite is shorter than C – C bond length in diamond – explain.
Answer:
In graphite each carbon is in sp² hybridisation and makes three sigma bonds with three neighbouring carbon atoms. Fourth electron forms a π bond. The electron is delocalised over the whole sheet. Due to this C – C bond length is shorter in graphite. In diamond each carbon is in sp³ hybridisation. So every carbon forms four C – C single bonds. sp²-sp² bond is stronger than sp³-sp³ bond. Hence C – C bond length in diamond is longer than in graphite.
Question 28.
Diamond is used as precious stone-explain.
Answer:
The refractive index of diamond is maximum 2.45. Due to this high refractive index when light falls on diamond total internal reflection takes place. So diamonds glitter in light. Hence they are used as precious stones.
Question 29.
Carbon never shows coordination number greater than four while other members of carbon family show coordination number as high as six – explain.
Answer:
In carbon the outermost orbit is second orbit which do not contain d-orbitals. So it can form only 4 bonds. In other elements, their atoms contain vacant d-orbitals in their valence shell. So they can accept electron pairs and can show coordination number 6. But carbon cannot exhibit a coordinate number more than 4.
Question 30.
Producer gas is less efficient fuel than water gas – explain.
Answer:
Producer gas contain about 33% CO and 65% N2. Nitrogen is not combustible. So the percent of cotnbustive gas in producer gas is less.
Water gas is a mixture of CO and H2 and both are combustible. So the percent of combustible gases in water gas is more.
Thus producer gas is less efficient than water.
Question 31.
SiF2-6 is known while SiCl2-6 is not-explain. [AP ’16]
Answer:
The main reasons are :
- Si+4 is small in size.
- It is not possible to accommodate six Cl– ions around a small Si+4 ion.
- The interaction of lone pairs on Cl– ion and Si+4 ion is weak.
Short Answer Questions
Question 1.
Explain the difference in properties of diamond and graphite on the basis of their structure.
Answer:
Differences between diamond and graphite
| Diamond | Graphite |
| 1. Carbon is in sp³ hybridised state. | 1. Carbon is in sp² hybridised state. |
| 2. It is a bad conductor of electricity. | 2. It is a good conductor of electricity. |
| 3. It has a giant three dimensional – polymeric network structure. | 3. It has a two dimensional layer structure. |
| 4. C-C bond length is 1.54 Å. | 4. C-C bond length is 1.42Å and distance between two layers is 3.4 Å. |
| 5. It is a hard substance. | 5. it is a soft substance. |
![]()
Question 2.
Explain the following.
a. PbCl2 reacts with Cl2 to give PbCl4.
b. PbCl4 is unstable to heat.
c. Lead is not known to form Pbl4.
Answer:
a. In PbCl2 lead is in +2 oxidation state. Chlorine is a good oxidising agent and can oxidise Pb2+ to Pb4+. So PbCl2 can react with Cl2 forming PbCl4.
b. Pb² is more stable than Pb4+ due to inert pair effect. So PbCl2 is more stable than PbCl4. Thus on heating PbCl4 converts into stable PbCl2 by losing Cl2.
c. Pb4+ is unstable due to inert pair effect and can easily convert into stable Pb2+ So Pb4+ can act as a good oxidising agent. T ion is a good reducing agent and reduces Pb4+ to stable Pb2+. So PbI4 is not formed.
Question 3.
Explain the following.
a. Silicon is heated with methyl chloride at high temperature in the presence of copper.
b. SiO2 is treated with HF
c. Graphite is a lubricant
d. Diamond is an abrasive
Answer:
a. When silicon is heated with methyl chloride at high temperature in the presence of copper various types of methyl substituted chlorosilanes are formed.

b. When SiO2 is treated with HF, H2SiF6 is formed
SiO2 + 4HF → SiF4 + 2H2O
SiF4 + 2HF → H2SiF6
c. Graphite has two – dimensional layer structure and these layers can slide easily one over the other. Hence graphite is used as a lubricant.
d. In diamond every carbon is in sp³ hybridisation and is in bond with four other carbon atoms tetrahedrally. The structure extends in space and produce a rigid three dimensional network of carbon atoms. In this structure directional covalent bonds are present throughout the lattice. To break these covalent bonds very high energy is required. Therefore diamond is the hardest substance and thus can be used as abrasive.
Question 4.
What do you understand by [AP Mar. ’19]
a. Allotropy
b. Inert pair effect
c. Catenation
Answer:
a. Allotropy :
The existence of an element in different physical forms but possessing similar chemical properties is known as allotropy.
The three crystalline allotropes of carbon are diamond, graphite and fullerenes.
b. Inert pair effect:
The reluctance of ns electrons to unpaired and take part in bond formation is called inert pair effect. Due to inert pair effect the stability of lower oxidation state increases while the stability of higher oxidation state decreases in a group from top to bottom.
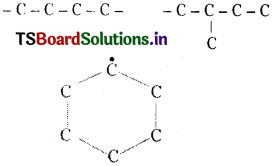
c. Catenation :
Combining capacity of the atoms of same element to form long chains, branched chains and cyclic compounds is called catenation. Of all the elements carbon has maximum catenation power.
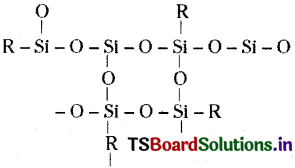
Question 5.
If the starting material for the manufacture of silicones is R SiCl3, write the structure of the product formed.
Answer:
R SiCl3 + 3H2O → R Si (OH)3
Condensation of the hydrolysed product give three dimensional silicone
Question 6.
Write a short note on zeolites.
Answer:
Zeolites are three dimensional alumino sili-catesrlf some silicon atoms in three dimensional network silicon dioxide are replaced by aluminium atoms, the alumino silicate formed will have some negative charge. To balance these negative charges some extra cations such as Na+, K+ or Ca2+ will present in these silicates. Examples are ZSM – 5 which is used as a catalyst in direct conversion of alcohols into gasoline. Hydrated zeolites are used as ion exchangers in softening of hard water.
The structure of zeolite permits the formation of cavities of different sizes. Molecules like H2O, NH3, CO2 can be trapped in these cavities. Thus they can serve as molecular sieves.
![]()
Question 7.
Write a short note on silicates.
Answer:
Silicates compounds are formed from the orthosilicic acid. All silicates have the basic structural unit SiO4-4 which has tetrahedral structure.
When discrete SiO4-4 unit is present in silicates they are called orthosilicates. Different silicates are formed by joining a number of SiO4-4 units through the corners of SiO4-4 tetrahedron by sharing 1, 2, 3 or 4 oxygen atoms per silicate unit. Thus chain, ring, sheet or three dimensional silicates are formed depending on the number of oxygen atoms shared. Negative charge on the silicate is neutralised by the positive charged metal ions.
Question 8.
What are silicones? How are they obtained?
Answer:
Silicones are organo silicon compounds containing (R2 Si – O) as repeating unit. First alkyl or aryl substituted chlorosilanes are prepared. These on hydrolysis followed by condensation, polymerisation gives polymeric silicones. Eg.
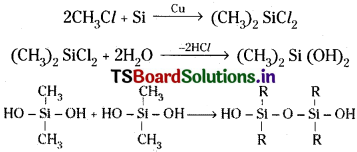
This reaction continue to give chain polymers. If water molecule is eliminated from the terminal OH groups of same chain cyclic, silicones are formed.
Question 9.
Write a short note on fullerene.
Answer:
Fullerene is a crystalline allotrope of carbon. Fullerenes are made by heating graphite in an electrie arc in the presence of inert gas. The soot formed by condensation of vapour of carbon contains mainly C60 fullerene molecules. It is known as Buckminster fullerene. It has soccer ball shape.
C60 fullerene contains 20 six membered rings and 12 five membered rings. A six membered ring is fused with either six membered ring or with five membered ring but a five membered ring is always fused with six membered ring. All carbon atoms in fullerene are in sp² hybridisation and are in bond with three other carbon atoms. The fourth electron of carbon delocalises on all carbon atoms giving aromatic character to fullerene. These are also called bucky balls, in short.
Question 10.
Why SiO2 does not dissolve in water?
Answer:
SiO2 has gaint polymeric network structure. In this structure directional covalent bonds are present throughout the lattice. Due to this structure the molecule is non-polar. Hence it is insoluble in water.
Question 11.
Why is diamond hard?
Answer:
In diamond every carbon is in sp³ hybridisation and is in bond with four other carbon atoms tetrahedrally. The structure extends in space and produce a rigid three dimensional network of carbon atoms. In this structure directional covalent bonds are present throughout the lattice. To break these covalent bonds very high energy is required. Therefore diamond is the hardest substance and thus can be used as abrasive.
Question 12.
What happens when the following are heated?
a. CaCO3
b. CaCO3 and SiO2
c. CaCO3 and excess of coke.
Answer:
a) CaCO3
CaCO3 decomposes to give CaO and CO2
![]()
b) CaCOs and SiO2
CaO formed after the decomposition of CaCO3 combine with SiO2 forming CaSiO3
CaCO3 → CaO + C02 ↑
CaO + SiO2 → CaSiO3
c) CaCO3 and excess of coke
First CaCO3 decomposes forming CaO and CO2. The CaO react with excess of carbon forming calcium carbide.
CaCO3 → CaO + CO2
CaO + 3C → CaC2 + CO
Question 13.
Why does Na2CO3 solution turn into a suspension, when saturated with CO2 gas?
Answer:
Sodium bicarbonate is sparingly soluble in water due to polymerisation of bicarbonate ions through hydrogen bonds. When CO2 is passed into sodium carbonate it converts into less soluble bicarbonate. So a suspension of sodium bicarbonate forms.
Na2CO3 + H2O + CO2 → 2 Na HCO3
![]()
Question 14.
What happens when
a. CO2 is passed through slaked lime
b. CaC2 is heated with N2
Answer:
a. CO2 is passed through slaked lime :
When CO2 is passed through slaked lime turns to milky due to the formation of insoluble CaCO3.
Ca(OH)2 + CO2 → CaCO3 + H2O + CO2
If excess CO2 is passed, the milkyness disappears due to the conversion of in-soluble CaCO3 to soluble Ca(HCO3)2.
CaCO3 + H2O + CO2 → Ca(HCO3)2
b. CaC2 is heated with N2:
On heating CaC2 with N2 calcium cynamide will be formed.
CaC2 + N2 → Ca CN2 + C (graphite)
The mixture calcium cynamide and graphite formed in this reaction is called nitrolim.
Question 15.
Write a note on the anomalous behaviour of carbon in the group-14.
Answer:
Carbon differs from its congeners in the following respects :
- Carbon occurs in free state whereas other elements are almost not available in free state.
- Carbon has no available d-orbitals in its valence shell while other elements have available d – orbitals.
- The maximum covalency of carbon is 4 whereas other elements exhibit a maximum covalency of 6.
- Carbon has a very high catenating ability.
- Carbon alone can form multiple bonds among themselves.
- Carbon forms a large number of hydrides known as hydrocarbons, which are thermally very stable. The other elements form limited number of hydrides which are thermally not very stable.
- Reducing nature of carbon is very high while the reducing nature of others is less.
- The halogen compounds of carbon are not hydrolysed while the other halogen compounds are readily hydrolysed.
Long Answer Questions
Question 1.
What are silicones? How are they pre-pared? Give one example. What are their uses?
Answer:
Silicones are organo silicon compounds containing (R2 Si – O) as repeating unit. First alkyl or aryl substituted chlorosilanes are prepared. These on hydrolysis followed by condensation, polymerisation gives polymeric silicones. Eg.

This reaction continue to give chain polymers. If water molecule is eliminated from the terminal OH groups of same chain cyclic, silicones are formed.
Uses of silicones :
- Used in the preparation of silicone rubbers.
- Used in the preparation of water-proof clothes and papers.
- Used in the preparation of grease, lubricants in aeroplanes.
- Used in electrical motors as insulators.
- Used in paints and enamels.
Question 2.
Explain the structure of silica. How does it react with a. NaOH and b. HF?
Answer:
Silica is gaint polymeric molecule. Every silicon in silica is in sp³ hybridisation. It is surrounded by 4 oxygen atoms tetrahedrally. Every oxygen is in bond with two silicon atoms.
a) Reaction with NaOH :
On heating with NaOH silica forms sodium silicate called water glass.
2 NaOH + SiO2 → Na2 SiO3 + H2O
b) Reaction with HF:
With HF silica forms SiF4 and H2SiF6
SiO2 + 4 HF → SiF4
SiF4 + 2HF → H2SiF6
Question 3.
Write a note on the allotropy of carbon.
Answer:
The existence of an element in different physical forms by possessing similar chemical properties is known as allotropy.
Carbon has many allotropic forms. They are of both crystalline and amorphous. The crystalline allotropes of carbon are diamond, graphite and fullerenes.
Diamond :
In diamond each carbon atom undergoes sp³ hybridisation and is in bond with four other carbon atoms in a tetrahedral form. This structure extends in space and produces a rigid three dimensional network of carbon atoms. These bonds are present throughout the lattice. To break all these extended bonds large amount of energy is required. So diamonds are very hard and have high melting point. They are used as precious stones, abrasives, cutting tools etc.
Graphite :
It has layered lattice structure in which every carbon is in sp² hybridisation and in bond with three carbon atoms. The carbon atoms are arranged in sheets containing hexagonal rings. The fourth electron is in re bond and delocalised on all the carbon atoms in a sheet. The distance between the layers is 340 pm. They are held together by van der Waals forces. They are slippery in nature. Hence graphite can be used as solid lubricant. Due to delocalised electrons graphite act as a good electrical conduction.
Fullerene :
Fullerene is a crystalline allotrope of carbon. Fullerenes are made by heating graphite in an electrie arc in the presence of inert gas. The soot formed by condensation of vapour of carbon contains mainly C60 fullerene molecules. It is known as Buckminster fullerene. It has soccer ball shape.
C60 fullerene contains 20 six membered rings and 12 five membered rings. A six membered ring is fused with either six membered ring or with five membered ring but a five membered ring is always fused with six membered ring. All carbon atoms in fullerene are in sp² hybridisation and are in bond with three other carbon atoms. The fourth electron of carbon delocalises on all carbon atoms giving aromatic character to fullerene. These are also called bucky balls, in short.
![]()
Question 4.
Write a note on
a. Silicates
b. Zeolites
c. Fullerenes
Answer:
a. Silicates:
Silicates compounds are formed from the orthosilicic acid. All silicates have the basic structural unit SiO4-4 which has tetrahedral structure.
When discrete SiO4-4 unit is present in silicates they are called orthosilicates. Different silicates are formed by joining a number of SiO4-4 units through the corners of SiO4-4 tetrahedron by sharing 1, 2, 3 or 4 oxygen atoms per silicate unit. Thus chain, ring, sheet or three dimensional silicates are formed depending on the number of oxygen atoms shared. Negative charge on the silicate is neutralised by the positive charged metal ions.
b. Zeolites:
Zeolites are three dimensional alumino sili-catesrlf some silicon atoms in three dimensional network silicon dioxide are replaced by aluminium atoms, the alumino silicate formed will have some negative charge. To balance these negative charges some extra cations such as Na+, K+ or Ca2+ will present in these silicates. Examples are ZSM – 5 which is used as a catalyst in direct conversion of alcohols into gasoline. Hydrated zeolites are used as ion exchangers in softening of hard water.
The structure of zeolite permits the formation of cavities of different sizes. Molecules like H2O, NH3, CO2 can be trapped in these cavities. Thus they can serve as molecular sieves.
c. Fullerenes :
Fullerene is a crystalline allotrope of carbon. Fullerenes are made by heating graphite in an electrie arc in the presence of inert gas. The soot formed by condensation of vapour of carbon contains mainly C60 fullerene molecules. It is known as Buckminsterfullerene. It has soccer ball shape.
C60 fullerene contains 20 six-membered rings and 12 five-membered rings. A six membered ring is fused with either six membered ring or with five membered ring but a five membered ring is always fused with six-membered ring. All carbon atoms in fullerene are in sp² hybridisation and are in bond with three other carbon atoms. The fourth electron of carbon delocalises on all carbon atoms giving aromatic character to fullerene. These are also called bucky balls, in short.